A fit-for-purpose pharmacy preparation
We help you develop a cost-effective dosage form that provides maximum dose flexibility to achieve your Phase I objectives.
Bridge formulations within the same FIH protocol
Evaluate formulation technologies and different dosage forms so you can select a patient-ready formulation to move forward.
Drug product to start your patient trials
Get immediate clinical trial material supply, ready for packaging and shipment, so you can start your Phase II proof-of-concept (POC) trials.
Begin your first-in-human testing with a simple, fit-for-purpose pharmacy preparation to get in the clinic quickly.
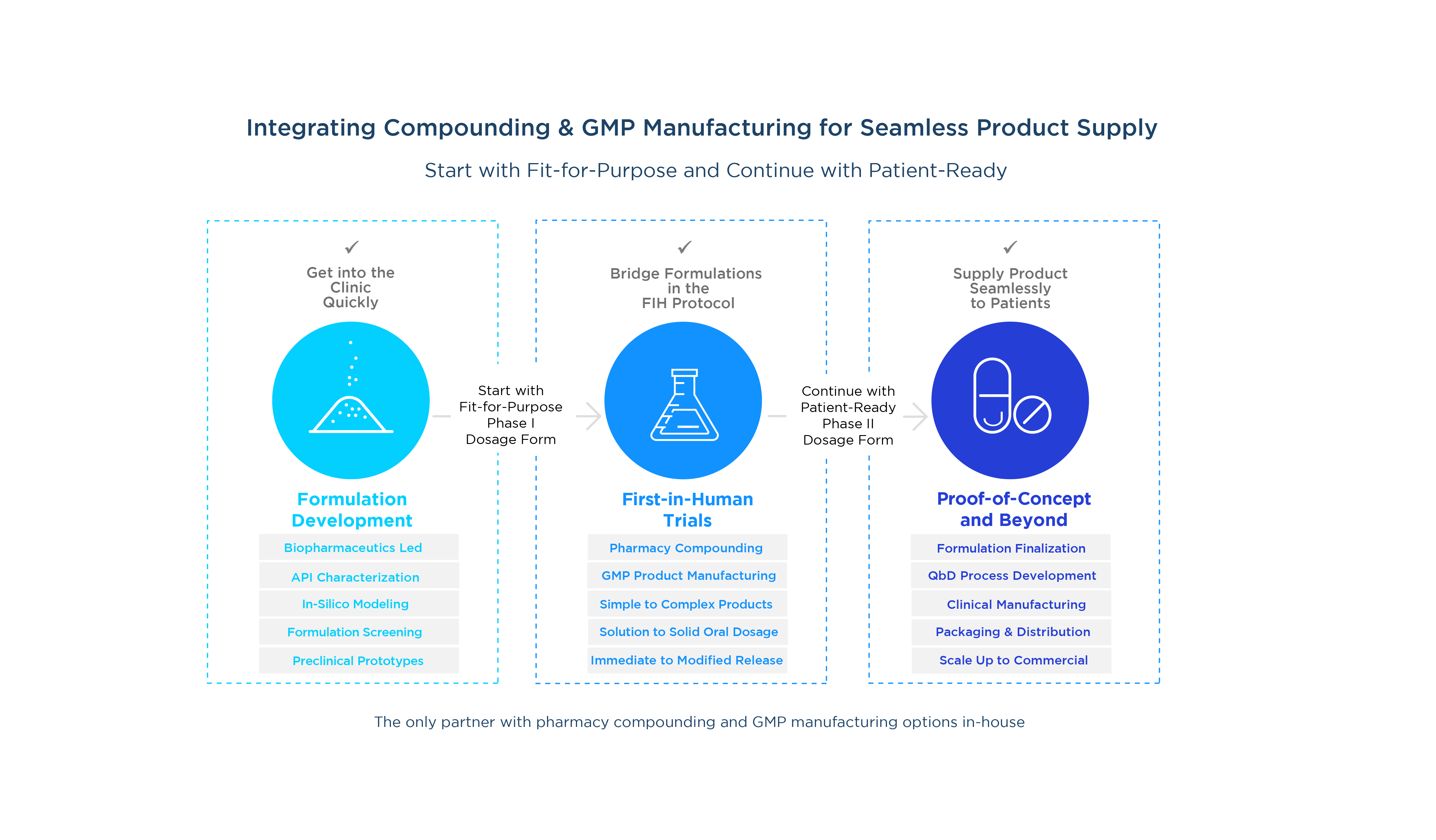
Later, advance from FIH to POC trials seamlessly
Our compounding pharmacy is fully integrated within our network of pharmaceutical development and GMP manufacturing facilities in the US and UK. Following your FIH clinical testing, we'll work with you to advance a patient-friendly GMP drug product into proof-of-concept trials as efficiently as possible.