Case Study: Pulmatrix
Breathing new life into Phase I clinical trials. Read Pulmatrix's case study.
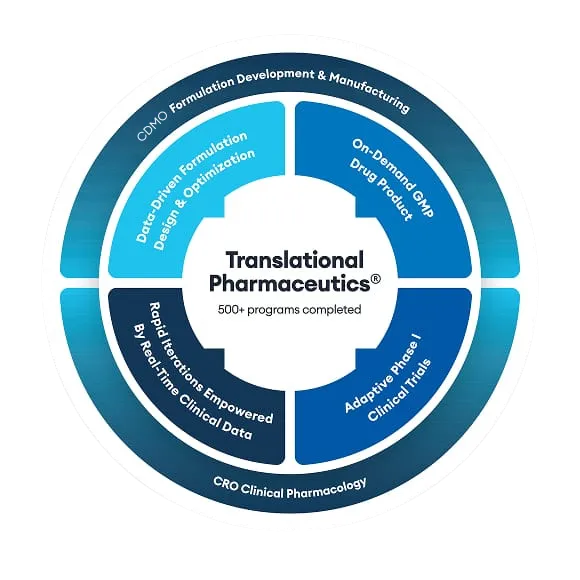
Pulmatrix is a small, clinical-stage biotechnology company based in Lexington, MA, that uses an innovative inhaled dry powder technology, iSPERSE (inhaled small particles easily respirable and emitted), to develop a new generation of inhaled therapeutics.
Initially drawn to Quotient Sciences' to perform a Phase I study based on our deep understanding of the UK regulatory environment, Pulmatrix realized that we are very experienced at designing flexible clinical trials across a broad formulation development space as well as having expertise in inhaled drug development. This was not something that they had come across in the US market which would prove to benefit their drug development timelines. By working with the same team of experts, they were able to streamline many processes and planning lessons allowing their drug program to progress more quickly.
Request a copy of our case study with Pulmatrix today and find out how we can accelerate your drug development timelines.