Discover the time savings and financial benefits of using Translational Pharmaceutics®, as evaluated by the Tufts Center for the Study of Drug Development (Tufts CSDD)
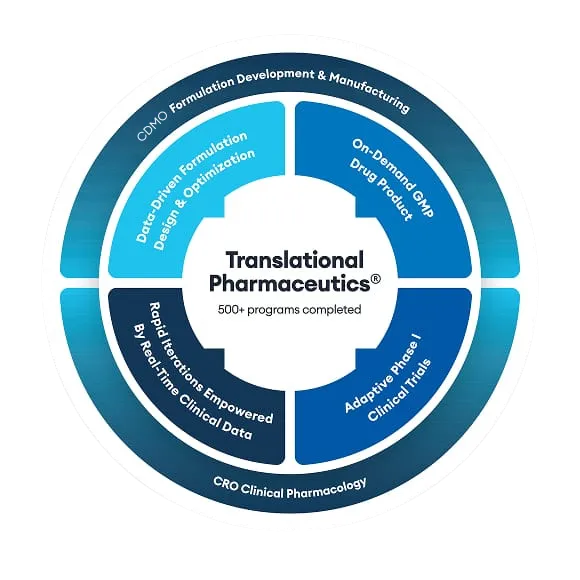
Quotient Sciences Translational Pharmaceutics® platform accelerates drug development by integrating services traditionally found from multiple contract research organizations (CROs), contract manufacturing organizations (CMOs), and other outsourcing partners.
For almost twenty years, it has been used widely by pharmaceutical and biotech companies, with more than 500 programs completed to advance molecules across the full development cycle.
How does Translational Pharmaceutics® enable faster development and greater savings? See what the Tufts Center for the Study of Drug Development (Tufts CSDD) has to say about the benefits of the Translational Pharmaceutics® platform for drug development when compared to traditional methods of development.
What can I learn about Translational Pharmaceutics® in the Tufts CSDD study?
Discover how your company can benefit from the savings and financial benefits that Translational Pharmaceutics® programs offer.

An accelerated path to pivotal drug development milestones

Average timeline savings of >12 months

R&D spend reduced by up to $9.5 million for each month of time saved

An integrated development model and simplified supply chain
Emerging biotech and Fortune 100 pharmaceutical clients have benefitted from applying Translational Pharmaceutics® platform to their drug development. See how it works in real customer case studies.