Nottingham, U.K., Dec. 11, 2018 – Quotient Sciences, a leading drug development services organization, and Druggability Technologies (DRGT), a specialty pharmaceutical company, announced a collaboration today to utilize Quotient Sciences’ integrated Translational Pharmaceutics® platform to advance the development of DRGT-46, a novel, fast-acting formulation of celecoxib using proprietary super-API compositions developed by DRGT. This will be Quotient Sciences first Translational Pharmaceutics® program delivered from its U.S. operating facilities.
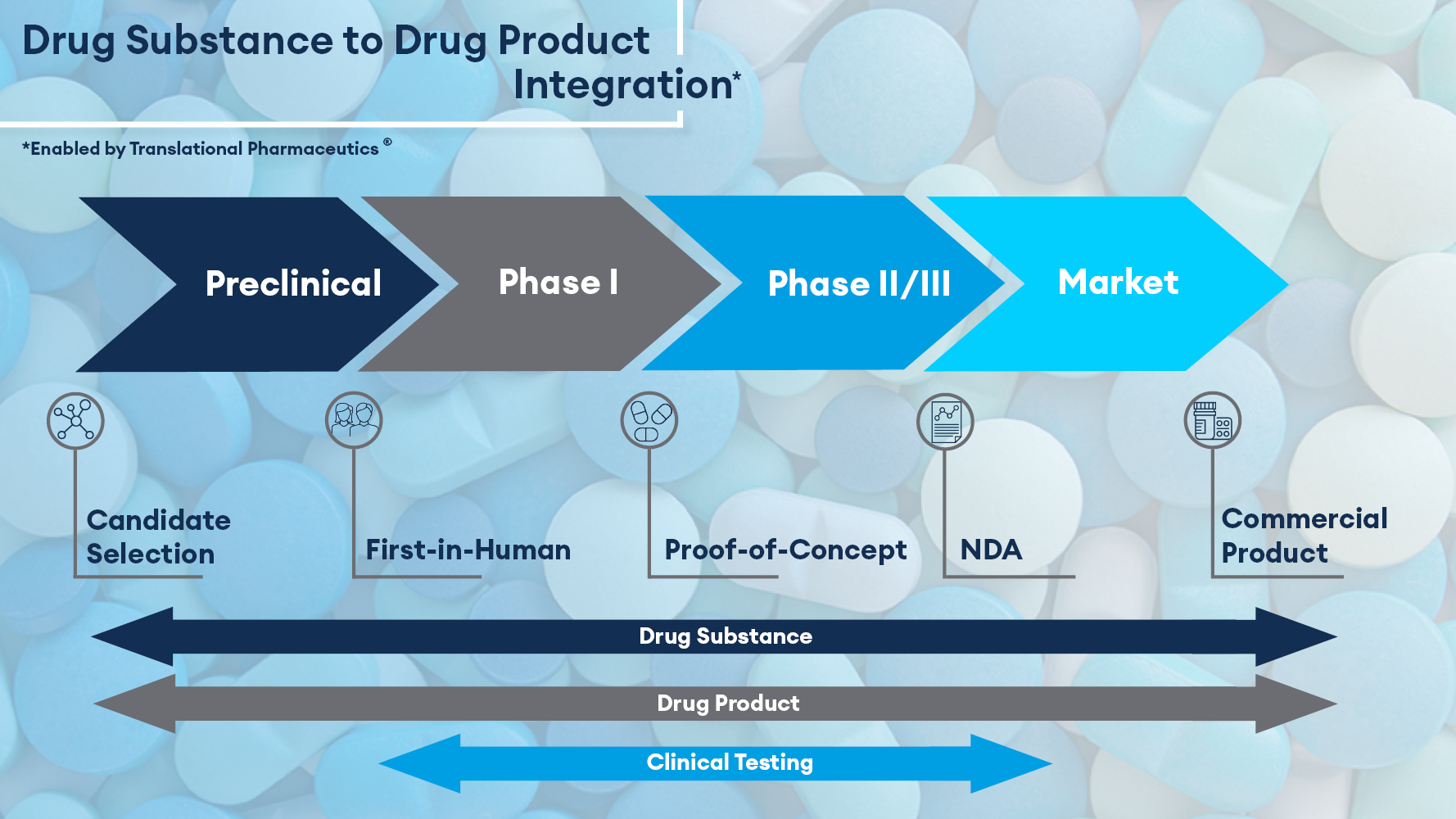
Within the program of work, DRGT is accessing Quotient Sciences' unique Translational Pharmaceutics® platform. This involves the development of amorphous spray-dried formulations and GMP clinical trial manufacturing in Philadelphia followed by clinical dosing and taste assessment at the Quotient Sciences clinical pharmacology unit in Miami. The program is fully adaptive, which will enable DRGT to optimize the drug product in response to emerging clinical data. Quotient Sciences will then seamlessly scale up the selected product to support pivotal patient trials.
Translational Pharmaceutics® transforms the traditional model of outsourcing to disparate contract CDMOs and CROs by integrating formulation development, real-time adaptive GMP manufacturing, and clinical research. It is proven to accelerate development timelines and reduce overall costs and has been applied in over 350 programs for customers worldwide. In 2017, Quotient Sciences established the capability to run Translational Pharmaceutics® programs in the U.S. Following a Critical Path Innovation Meeting with the FDA’s Office of Translational Sciences, and meetings with local institutional review boards, Quotient is now delivering programs in the U.S.
"We have significantly expanded our CDMO and CRO operations in the U.S. and are now performing integrated programs in the world’s largest R&D market," said Mark Egerton, Ph.D., CEO of Quotient Sciences. “This offers significant advantages to our customers. We are delighted to support DRGT in accelerating DRGT-46 as a new treatment for acute and chronic pain.”
Gábor Heltovics, CEO of DRGT, added, “We are very pleased to be working with Quotient Sciences on this accelerated development project with our proprietary super-API technology. The Company's tight integration of formulation, manufacturing and clinical research services has enabled DRGT to execute an efficient development plan with a single outsourcing partner and, most importantly, reduce our time to market.”
Quotient Sciences' comprehensive drug product expertise spans all types of oral dosage forms, a broad range of solubilization technologies, and high potency handling capabilities.